In his letter of 20 March 1800 to Sir Joseph Banks, Volta describes, alongside the column battery, another configuration of his artificial electric organ: the cup battery. This version highlights the role of liquids and metals in generating continuous electricity.

The device consists of a series of non-metallic cups or glasses, preferably made of glass, filled halfway with pure water or, better still, salt water or alkaline solutions. The cups are arranged in succession and connected to each other to form a real electrical chain.
The connection between one cup and the next is made using metal arches composed of two different metals. One end, immersed in a cup, is made of copper, while the other, immersed in the next cup, is made of tin or, preferably, zinc. The two metals are welded together above the liquid level, ensuring good electrical contact.
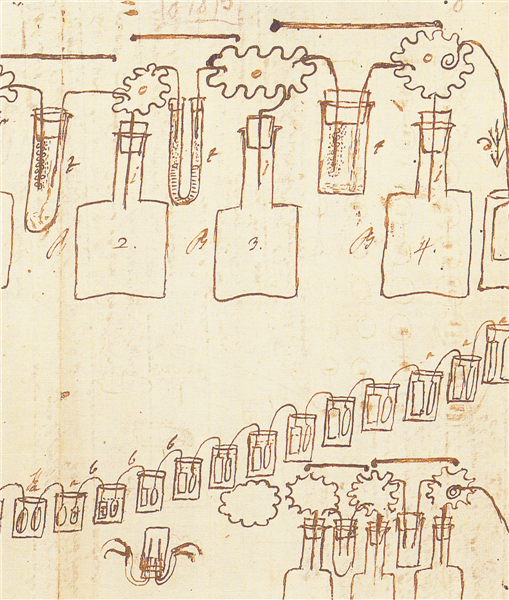
Volta observes that the choice of liquid affects the effectiveness of the device: alkaline solutions are more suitable when using tin, while salt water is preferable in the presence of zinc. The liquid acts as a second-class conductor, completing the electrical action between the metals.
Volta demonstrates that the overall effect produced by the succession of metals and liquids is equivalent to that which would result from direct contact between the first and last metals in the chain. This observation confirms that electrical force depends on the nature of the extreme metals and not on the intermediate contacts, a fundamental principle of contact electricity.
