After discovering the flammable air in the marshes near Angera, Volta observed that a mixture of flammable air and ordinary air, when subjected to an electric discharge, produced an explosion. To study this phenomenon quantitatively, he built a specific instrument: the eudiometer.

The eudiometer consists of a glass tube, with one end immersed in a basin of water and the other closed by a sealed cork stopper. Two metal wires pass through the stopper and terminate inside with electrodes, designed to produce the electric spark.

After filling the tube with a mixture of ordinary air and flammable air, Volta strikes a spark between the electrodes. The resulting explosion causes the water level in the tube to rise, indicating that some of the air has disappeared during the reaction.
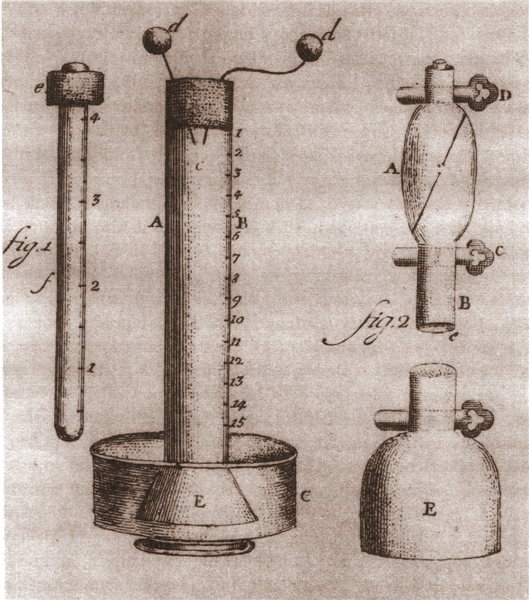
Volta interprets the result as the disappearance of flammable air and part of the common air, leaving “phlogisticated” air, i.e. air devoid of oxygen, in the tube. These observations were made at a time when the composition of air was not yet known.
In 1777, Volta communicated his findings to Joseph Priestley and continued his research with hydrogen and oxygen. In 1782, he presented these experiments in Paris to Lavoisier, who would go on to discover the chemical composition of water the following year.

Durante le combustioni Volta osserva spesso la comparsa di una rugiada sulle pareti del tubo, senza però riconoscerla come acqua. L’uso dell’acqua nell’eudiometro gli impedisce di accorgersi della sintesi dell’acqua, che sarà invece chiaramente individuata da Lavoisier ripetendo gli esperimenti con mercurio al posto dell’acqua.
